Designed by CHAIRONE

-
AbClon Inc. #1401 285, Digital-ro, Guro-gu, Seoul, Republic of Korea
T. +82-2109-1294
F. +82-2109-1296
E. abclon@abclon.com

CAR-T therapy is a type of immunotherapy where patient’s own immune cells are genetically engineered to specifically target cancer cells.

AbClon’s proprietary zCAR-T treatment is the next generation of CAR-T treatment whose modularity
can allows its to be used against various cancer targets as well modulating the activity of CAR-T cells within the patient.
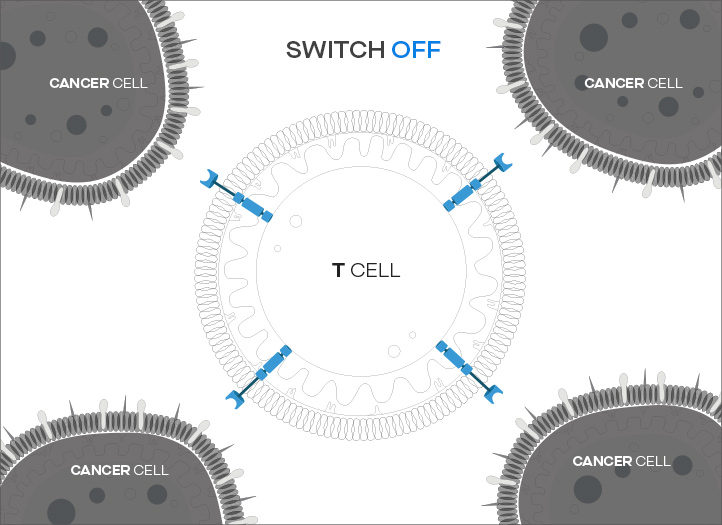
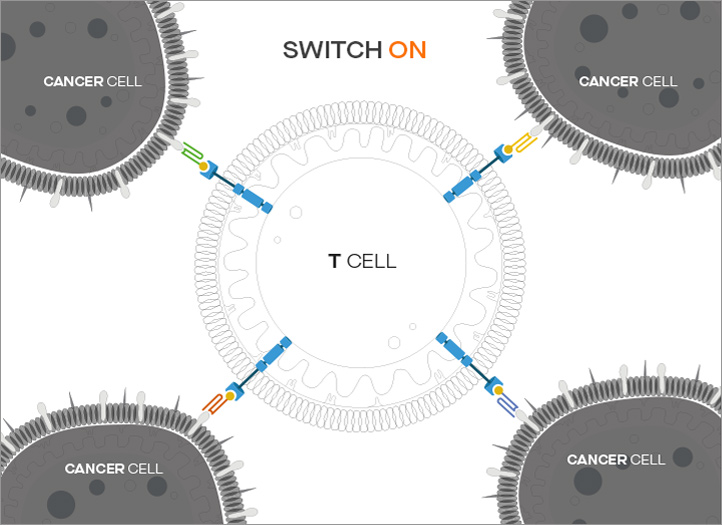
 ON/OFF function
ON/OFF function
 Controllable level of
Controllable level of  Expansion of
Expansion of